IGCSE Physics Past Papers Exam Questions (Edexcel) 2024 on Solids, Liquids and Gases
Use these IGCSE Physics Past Paper Exam Questions on the topic, Solid, Liquids and Gases to familiarise, practice and prepare for your IGCSE Physics examination.
You can find earlier questions in the same topic below:
- IGCSE Physics Past Year Exam Questions: Solids, liquids and gases 2019 – 2022
- IGCSE Physics Past Year Exam Questions: Solids, liquids and gases 2023 – 2024
What you need to know
Use the list below as a quick recap for what you need to know before attempting the past year exam questions under this topic. This is based on Edexcel International GCSE in Physics (4PH1) specification with first teaching Sept 2017 and first examination June 2019.
Paper 1 and 2: (5) Solids, liquids and gases
Paper 1 covers all the topics except where it is marked “Paper 2 only” while Paper 2 covers all topics.
A. Units
- degree Celsius (°C), Kelvin (K), joule (J), kilogram (kg), kilogram/metre3 (kg/m3), metre (m), metre2 (m2), metre3 (m3), metre/second (m/s), metre/second2 (m/s2), newton (N) and pascal (Pa)
- use the following unit: joules/kilogram degree Celsius (J/kg °C)
B. Density and pressure
- relationship between density, mass and volume. ρ = m/V
- investigate density using direct measurements of mass and volume.
- relationship between pressure, force and area. p = F/A
- pressure at a point in gas or liquid acts equally in all directions.
- relationship between pressure difference, height, density and gravitational field strength. p=h×ρ×g
C. Change of state (Paper 2 only)
- that heating a system will change the energy stored in that system. (raise its temperature or product changes of state)
- changes that occur when a solid melts to liquid and when a liquid boils or evaporates to gas.
- describe the arrangements of atoms in solids, liquids and gases.
- temperature-time graph to show a constant temperature during a change of state.
- specific heat capacity of a substance is the energy required to change the temperature of 1kg of a substance by 1°C. (J/kg °C)
- equation relating to the change in thermal energy to mass, specific heat capacity and temperature change. ΔQ = m × c × ΔT
- investigate the specific heat capacity of materials including water.
D. Ideal gas molecules
- how molecules in a gas have random motion and exert a pressure on the sides of their container
- understand why there is absolute zero at -273°C.
- describe Kelvin scale of temperature and convert between Kelvin and Celsius scales.
- increase in temperature results in increase in average speed of molecules.
- Kelvin temperatures of a gas are directly proportional to the average kinetic energies of its molecules.
- explain for a fixed mass of mass, the qualitative relationship between:
- pressure and volume at constant temperature;
- pressure and kelvin temperature at constant volume.
- the relationship between the pressure and kelvin temperature of a fixed mass of gas at constant volume. P1/T1 = P2/T2
- relationship between pressure and volume of a fixed mass of mass at constant temperature. P1V1=P2V2
June 2024 Paper 1P Q8
9 This question is about air pressure.
(a) During an aeroplane flight, a passenger drinks some water from a plastic bottle.
The passenger then replaces the top to seal the bottle, as shown in diagram 1.

The air pressure outside the bottle is 80 kPa.
State the air pressure inside the bottle just after the bottle has been sealed. (1)
air pressure = …………………………………………………….. kPa
(b) As the aeroplane descends, the air pressure inside the aeroplane changes.
When the aeroplane lands, the passenger notices that the plastic bottle has collapsed, as shown in diagram 2.

Explain why the bottle has collapsed. (2)
(c) Explain how gas molecules in the air exert a pressure on the surface of the bottle. (3)
(Total for Question 9 = 6 marks)
June 2024 Paper 1PR Q5
5 A student needs to determine the density of some small rocks that appear to all be made of the same material.

(a) The student decides to measure the mass and the volume of each rock.
Describe a method the student could use to accurately determine the mass and the volume of each rock.
You may draw a diagram to help your answer. (5)
(b) The table shows the student’s results for three of the rocks.
| Rock | Mass in g | Volume in cm3 |
| A | 15 | 6.3 |
| B | 13 | 6.1 |
| C | 16 | 7.5 |
(i) State the formula linking density, mass and volume. (1)
(ii) After looking at the data, the student concludes that one of the rocks may be made of a different material from the others.
Using the data from the table, justify the student’s conclusion. (4)
(Total for Question 5 = 10 marks)
June 2024 Paper 1PR Q11
11 This question is about gas pressure.
(a) Diagram 1 shows some of the molecules of a gas in a sealed container.
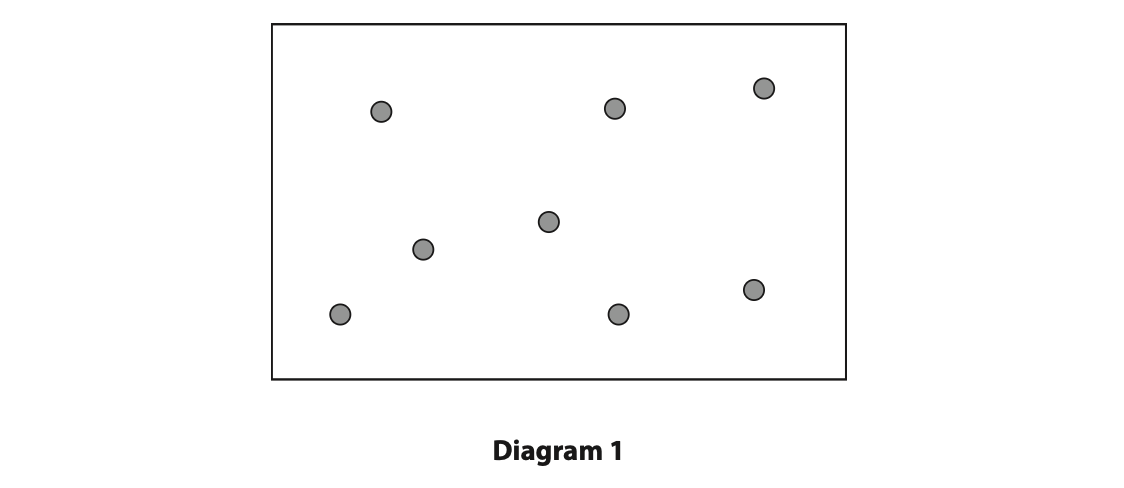
The molecules collide with all the surfaces of the container. This exerts an outward force on the container and causes pressure.
Describe how the motion of the gas molecules causes an equal pressure on all the walls of the container.
You may add to diagram 1 to help your answer. (2)
(b) The width of the container is slowly decreased so that the volume of the container is smaller than before.
Diagram 2 shows the width of the container before and after this change. All other dimensions of the container remain the same.
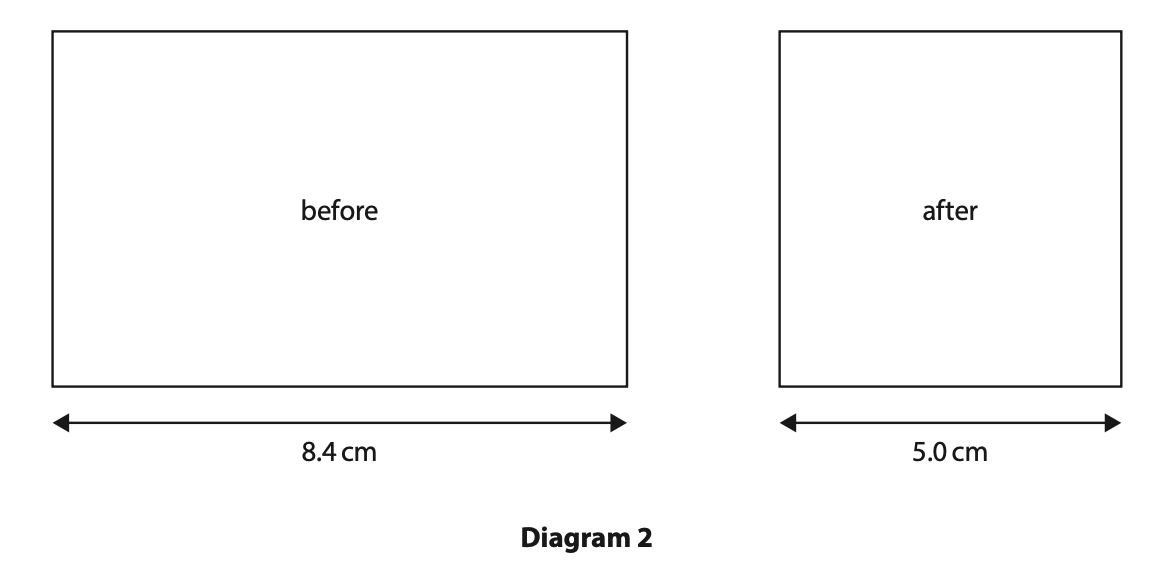
The initial volume of the gas is 130 cm3
The initial pressure of the gas is 100 kPa.
Calculate the pressure of the gas after the width of the container is decreased.
Assume the temperature of the gas remains constant. (4)
pressure = …………………………………………………….. kPa
(Total for Question 11 = 6 marks)
June 2024 Paper 2PR Q3
3 A sample of liquid gallium is allowed to cool in a laboratory.
The liquid gallium freezes to become a solid.
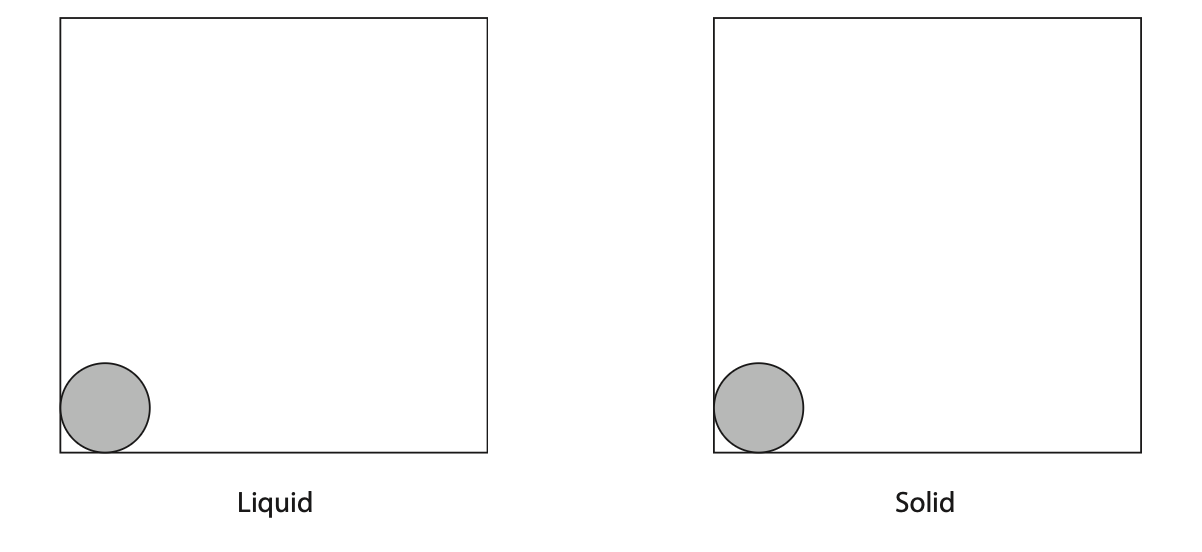
(a) Complete the diagram by drawing the arrangement of particles in a liquid and the arrangement of particles in a solid.
The first particle in each box has been drawn for you.
(b) The initial temperature of the sample of liquid gallium is 80 °C.
The freezing temperature of gallium is 30 °C.
The final temperature of the solid gallium is 20 °C.
Complete the graph to show how the temperature of the gallium changes during the time that it cools to 20 °C.
Add appropriate values to the temperature axis. (3)

November 2024 Paper 1P Q5
5 Diagram 1 shows the apparatus a student uses to determine the specific heat capacity of a liquid.
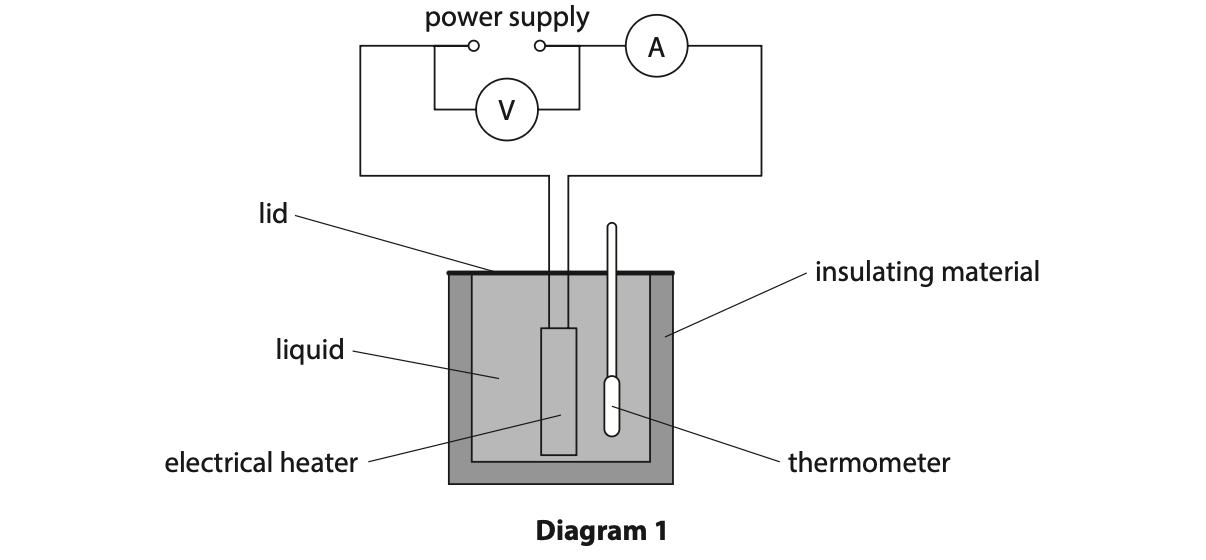
(a) Describe a suitable method the student could use to determine the specific heat
capacity of the liquid. (5)
(b) The graph shows the student’s results.
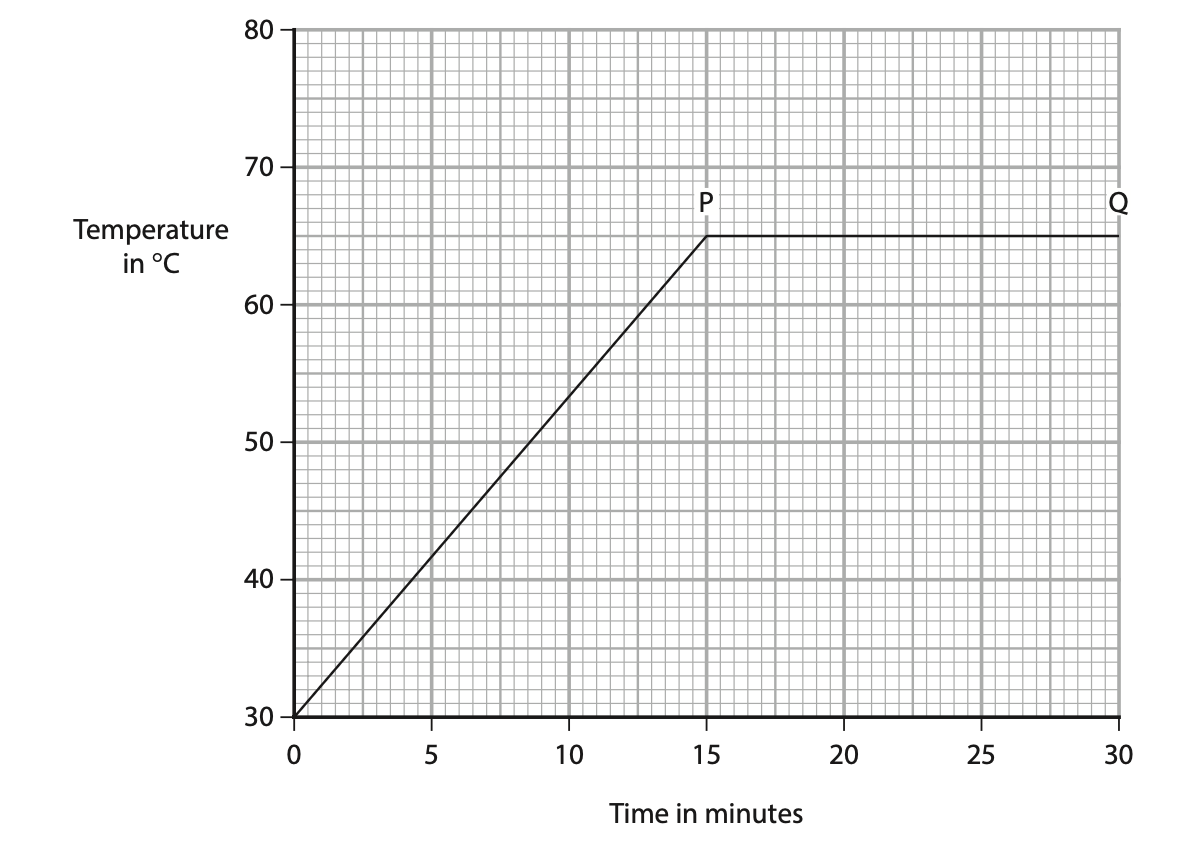
(i) What is happening to the liquid between points P and Q on the graph?(1)
A melting
B freezing
C boiling
D condensing
(ii) Use the graph to determine the change in temperature of the liquid after 15 minutes of heating. (1)
Temperature difference = …………………………………………………….. °C
(iii) The power of the heater is 48 W and the mass of the liquid is 0.53 kg.
Calculate the specific heat capacity of the liquid. (3)
specific heat capacity = …………………………………………………….. J / kg °C
(Total for Question 5 = 10 marks)
November 2024 Paper 1P Q7
(Forces and Motion + Solids, Liquids and Gases)
7 The diagram shows a submarine at rest underwater.
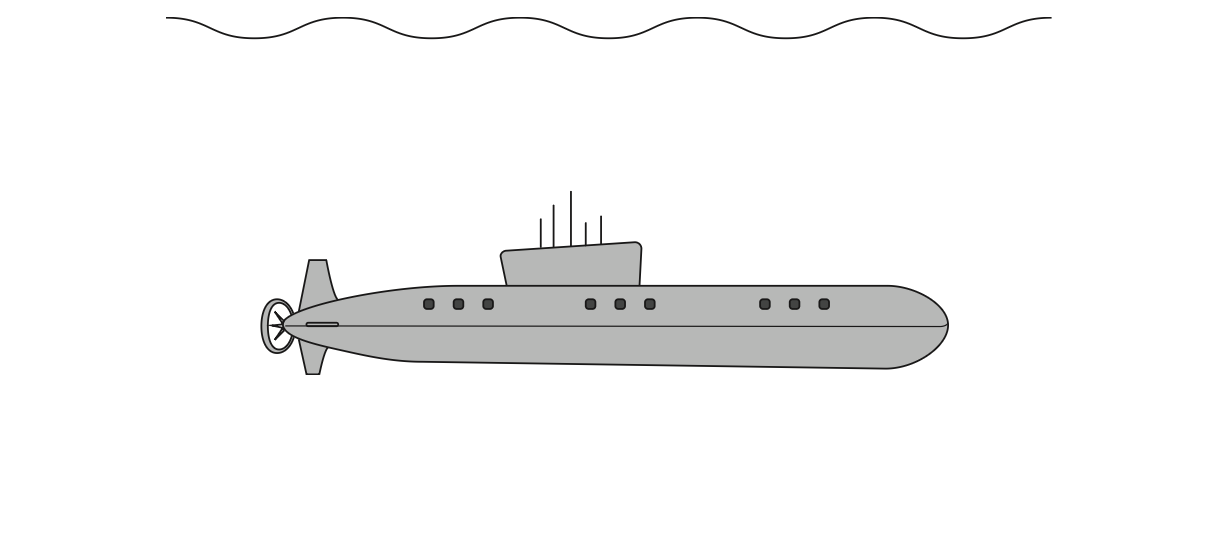
(a) There are two vertical forces acting on the submarine.
(i) One of the forces is called upthrust.
Give the name of the other force. (1)
(ii) Draw two arrows on the diagram to represent the vertical forces acting on the submarine. (2)
(b) (i) State the formula linking pressure, density, gravitational field strength and height. (1)
(ii) Calculate the pressure from the water at a point 38 m below the surface of the water. (2)
[density of sea water = 1030 kg/m3]
pressure = …………………………………………………….. kPa
(c) To rise back to the surface, air is pumped into a storage tank inside the submarine.
(i) Explain how the air exerts pressure on the walls of the storage tank. (3)
(ii) The air in the storage tank starts at a pressure of 410 kPa.
As the submarine rises, the air in the storage tank increases in temperature from 2.5°C to 18°C. The volume of the storage tank remains constant.
Calculate the pressure of the air in the storage tank at 18°C. (4)
pressure = …………………………………………………….. kPa
(Total for Question 7 = 13 marks)







